Organo-f-synthesis
Carbon Dioxide Removal by Direct Air capture
The aim is to capture carbon dioxide directly from the atmosphere at low concentrations (423 ppm), selectively, from other atmospheric constituents, which represents an initial scientific breakthrough. In this field, we manufacture innovative materials enabling selective capture. Once captured, the carbon dioxide must be efficiently released (outside the atmosphere) to regenerate the material, after which it can continue atmospheric capture. A second scientific bottleneck lies in the energy required for this operation, making current technologies uneconomical from an energy point of view. We propose to explore the use of plasmas in collaboration with Olivier Guaitella from the Plasma Physics Lab to promote release at a much lower energy cost than current thermal methods. The project is led by Grégory Nocton with Olivier Guaitella, Grégory Danoun, and Thomas Simler, and is supported by the foundation of École polytechnique.
Carbon dioxide valorization in organometallic chemistry
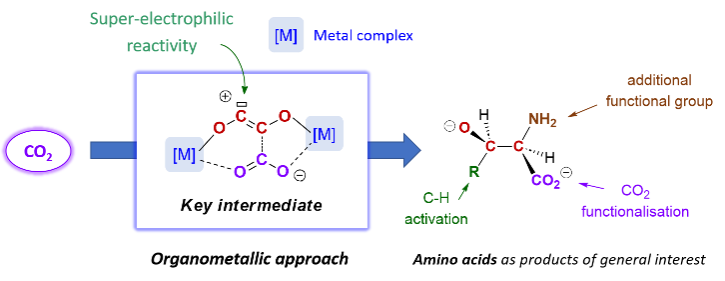
The group is using carbon dioxide as a C1 synthon for the formation of value-added products. Using an organometallic approach, we are transforming this inert and stable molecule into reactive intermediates, allowing further functionalization.
An example is the synergistic functionalization strategy (picture), which allows both the transformation of carbon dioxide and the activation of other small molecules, including alkanes. This original reactivity of carbon dioxide opens new avenues for its upgrading into elaborate scaffolds, such as amino acid motifs (Thomas, Simler, ANR JCJC MultiCO 2023-2026). A second approach concerns the photochemical Ln-OCO bond cleavage on high-valent lanthanide complexes for possible catalytic recycling.
Chem. Sci., 2022, 13, 7449-7461
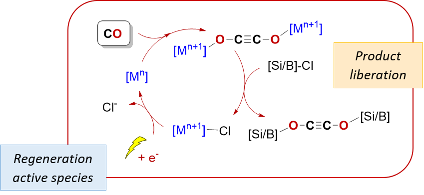
Electrochemical conversion of CO into high value-added molecules
Carbon monoxide is a useful synthon for the industrial synthesis of C1 oxygenates such as methanol or acetic acid, as well as for the formation of synthetic fuels. In contrast, processes allowing the controlled formation of multicarbon oxygenated products from CO are relatively scarce. In this project, we use low-valent oxophilic metal complexes to induce the reductive coupling of CO. The molecule obtained from this reaction is then a platform for further functionalization into value-added products. We are especially investigating the use of electrochemical methods to regenerate the low-valent metal complexes in order to go towards catalytic applications.
This project is led by Thomas Simler and has received funding from the “Young Researcher Program” of the ANR (ANR JCJC MultiCO 2023-2026).